
AN AUTHORITATIVE RESOURCE FOR EVERYTHING ABOUT CANCER
The Cervical Cancer Paradox: Disparities and Implementing Strategies in South Asia
Research Intern
Implementation Science & Research Development Officer

The Cervical Cancer Paradox: Disparities and Implementing Strategies in South Asia
Cervical cancer is one of the most preventable cancers, yet it continues to claim hundreds of thousands of lives each year, especially in South Asia. Despite advances in HPV vaccination and screening, systemic barriers, low screening uptake, and sociocultural challenges have created a persistent global disparity. This article explores the “cervical cancer paradox” and highlights scalable, community-driven solutions that can bridge the gap and accelerate progress toward elimination.
Introduction: Understanding the Cervical Cancer Paradox
The global oncology landscape is currently defined by a paradox; while cervical cancer remains nearly 93% preventable through human papillomavirus (HPV) vaccination and screening, the disease persists as a primary driver of mortality in resource-limited settings. Every year, healthcare professionals diagnose over 660,000 new cases, leading to approximately 350,000 deaths worldwide.¹ Low- and middle-income countries (LMICs) bear the brunt of this burden, accounting for nearly 90% of these global fatalities.²
This profound disparity transcends biological prevalence, serving as a stark indicator of systemic failures in preventive care delivery, entrenched sociocultural barriers, and a lack of accessible healthcare infrastructure.³ While cervical cancer has evolved into a manageable public health challenge in high-income nations such as the US, the UK, Australia, and Japan, LMICs like India and Nepal continue to face an epidemiological crisis.⁴ In response, the World Health Organization (WHO) has established the ambitious "90-70-90" targets for 2030, mandating that 90% of girls be fully vaccinated with the HPV vaccine by age 15, 70% of women be screened with a high-performance test by ages 35 and 45, and 90% of women identified with cervical disease receive treatment.¹ Realizing these objectives necessitates a strategic pivot from centralized, high-cost care delivery models toward decentralized, community-led, and technologically appropriate solutions.⁵
Regional Disparities in Cervical Cancer Outcomes
The disparity in clinical outcomes between high-income regions and South Asia is fundamentally anchored in a pervasive "screening gap."⁶ While screening coverage reaches 75–78% in the US and Australia and 69% in the UK, even Japan’s 44% uptake, one of the lowest in high-income countries, significantly outpaces the <8% participation metrics across South Asia.7,8
India’s Screening Crisis and Policy Response
India's National Family Health Survey 5 (NFHS-5) highlights this epidemiological crisis, showing that less than 2% of women undergo regular screenings across the country.9 This deficit persists even within high-literacy states such as Kerala, where coverage is at a mere 3.5%. Conversely, the state of Tamil Nadu has achieved superior screening benchmarks through the strategic integration of nurse-led Visual Inspection with Acetic Acid (VIA) and the implementation of high-impact media health literacy campaigns.10
Nepal’s Progress and Persistent Gaps
Nepal presents a similarly challenging landscape, where cervical cancer persists as a major component in cancer-related mortality among the female population.11 As of 2019, national screening coverage remained dismally low, with only 8.2% of women aged 30–49 having ever undergone screening, contributing to a staggering case fatality rate (CFR) of approximately 66%.12 This reality dictates that two out of every three women diagnosed will succumb to the malignancy, a tragic outcome primarily precipitated by advanced-stage clinical presentation.⁴
Table 1. Comparative Regional Burden and Screening Metrics (NIH/IARC, 2022-23)12
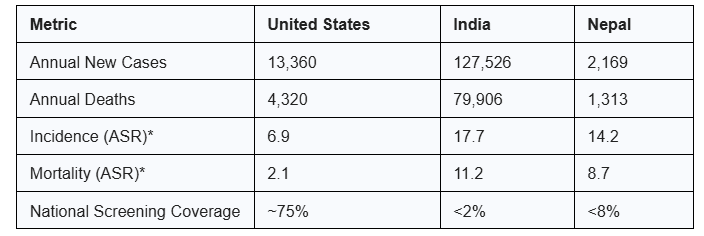
*ASR: Age-standardized rate per 100,000 women.
Recent epidemiological advancements offer a nascent paradigm of hope. In February 2026, the Government of India inaugurated an extensive national HPV immunization initiative, targeting 1.15 crore girls aged 14, with a 90-day intensive drive and a single-dose regime of Gardasil-4 or Cervavac.⁴ Data released during the rollout indicates that access is still heavily dictated by school type, family employment, and historical distrust.13 Concurrently, Nepal achieved a significant landmark in February 2025 by vaccinating over 1.46 million girls, attaining a 94% national administrative coverage that surpassed the initial 90% target five years ahead of the WHO schedule.14 However, these overall achievements hide important differences between regions; even though the national average is high, areas like Madhesh Province and the Kathmandu Valley only had coverage of 70% and 75%, respectively. These gaps, driven by high internal migration, frequent cross-border movement, and the growth of urban slums, indicate that while political commitment to primary prevention has been revitalized, the South Asian landscape still faces significant challenges in achieving granular health equity.
Innovative Strategies to Expand Screening and Prevention
To build sustainable cancer care in resource-limited settings, it is vital to adopt transformative innovations that overcome the logistical and financial hurdles of traditional models used in high-income countries/regions.
Point-of-Care Diagnostics and Self-Sampling
A primary element inf the strategies is the use of isothermal point-of-care (POC) diagnostic platforms, Key technologies include transcription-mediated amplification (TMA) utilized by the Aptima HPV Assay, Cross-Priming Amplification (CPA) in the EasyNAT HPV System, and various Loop-mediated isothermal amplification (LAMP) assays.15 Specialized isothermal platforms such as AmpFire and ScreenFire further facilitate decentralized screening by eliminating the need for complex thermal cycling infrastructure. Isothermal amplification utilizes portable, battery-operated devices that maintain a constant temperature. These molecular assays typically cost from US$5-15 per unit in low-resource settings. If combined with the utilization of self-collected vaginal swabs, which enhances screening participation in rural regions, particularly in LMICs, this approach will further mitigate barriers to screening. The strategies provide a culturally acceptable alternative that bypasses the logistical and psychosocial hurdles of provider-performed pelvic examinations.16
Screen-and-Treat Models to Reduce Loss to Follow-Up
Further, "Screen-and-treat" protocols leverage the rapid turnaround of isothermal POC assays to enable same-day clinical intervention, effectively collapsing the traditional multi-visit diagnostic pathway into a single encounter. By providing immediate results, these strategies minimize loss to follow-up, a critical challenge in LMICs where distance and cost often prevent patients from returning for subsequent treatment.
Patient Navigation and Virtual Tumor Boards
Complementing these technological innovations is the strategic deployment of the "Patient Navigator" model, a clinical framework recalibrated from high-income healthcare systems to effectively mitigate "loss to follow-up."17 Navigators help coordinate between grassroots health workers and tertiary care centers, supporting patients to confront a wide range of non-clinical issues, such as transportation, family responsibilities, and language barriers. High-level evidence indicates that patients receiving these services are 1.62 and 1.63 times more likely to complete initial screening and adhere to follow-up diagnostic resolution, respectively, compared to those receiving standard care.18
Setting up virtual tumor boards to fill important gaps in specialized clinical knowledge is another critical strategy, which Binaytara has initiated. These weekly sessions enable healthcare providers in Janakpur, Nepal, to engage in real-time consultations with world-renowned oncology specialists, including those from the Mayo Clinic and Stanford Medical Center.19 This collaborative model eliminates the necessity for patients in remote regions to engage in arduous international travel for specialized care by facilitating access to global clinical standards and evidence-based protocols.
Data and Evidence Supporting Community-Based Interventions
The Community-Based Intervention for Cervical Cancer Screening Uptake (COBIN-C) trial in Nepal rigorously validates the efficacy of these decentralized, community-led paradigms.⁵ This cluster-randomized investigation evaluated the impact of mobilizing Female Community Health Volunteers (FCHVs) to deliver home-based health literacy and catalyze screening participation.⁹
Table 2. COBIN-C Trial Outcome Data
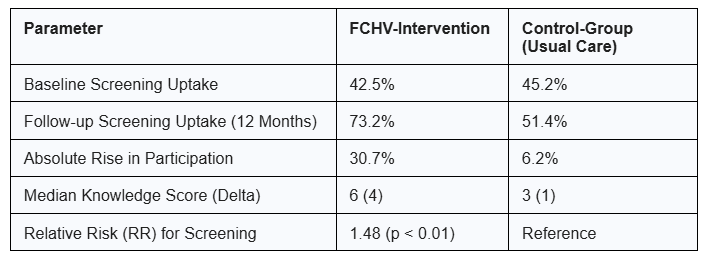
Data from the COBIN-C trial (Table 2) indicates that participants in the intervention group were 48% more likely to adhere to screening protocols than those in the control group. The study further confirmed the scalability and cultural relevance of utilizing community volunteers for outreach, as median health literacy scores regarding the causes and prevention of cervical cancer rose from 2 to 6, a substantial improvement.⁵
Macroeconomic forecasts highlight the importance of these measures. By 2050, meeting the WHO’s "90-70-90" targets could contribute to an estimated US$28 billion to the global economy by ensuring women remain active participants in the workforce.20
Specifically in India, implementing a homegrown single-dose vaccine with 90% coverage has the potential to reduce the prevalence of HPV 16 and 18 by 97% over the next 50 years.21 According to these projections, sustained investment in preventive health could prevent over 62 million global deaths by the year 2120.¹
Expert Insights on Achieving Cervical Cancer Elimination
Expert consensus in The Lancet Oncology frames cervical cancer elimination as a challenge of "political will rather than biological limitation."22 Recent health systems analyses identify Universal Health Coverage and nursing workforce density as the primary predictors of improved mortality-to-incidence ratios in low-resource settings.23 Gender inequality in cancer preventive care serves as an independent driver of cancer mortality; countries with higher disparity indices exhibit significantly worse clinical outcomes regardless of national wealth.23
To bridge the pervasive "know-do gap," many experts have heavily turned to implementation science, the systematic study of integrating evidence-based research into routine practice.24 This change shifts the global focus from the "what" (vaccines and screening) to the "how" (locally adapted delivery). Experts advocate for "screen-and-treat" protocols using portable, battery-operated technologies to bypass high-cost laboratory infrastructure.25 By prioritizing gender-responsive policies and decentralized care, achieving the WHO targets are feasible, ensuring that therapeutic breakthroughs reach the most vulnerable populations rather than bypassing them.26
Patient Perspectives: Breaking Cultural and Social Barriers
The patient experience across South Asia is often defined by a pervasive "culture of silence" and deep-seated sociocultural stigma.⁷ Many women in rural Nepal and India correlate reproductive health screenings with sexual promiscuity, fearing that a positive diagnostic result may precipitate accusations of infidelity or social ostracization.⁷ Furthermore, a systemic lack of female autonomy necessitates that many women obtain familial consent before accessing clinical services.⁵
However, the human dimension of oncology reveals remarkable resilience when catalyzed by health education. In one rural Nepali cohort, baseline literacy was alarmingly low, with only 6% of women familiar with cervical cancer and none cognizant of the Pap test.⁷
While initial testing acceptance was merely 15.6%, a 90-minute community-led education session resulted in 100% participation,⁷ underscoring a transformative "ripple effect": when a woman is empowered through screening and treatment, the dividends extend to her children, her family's financial stability, and the overall health of the community.⁴
Key Takeaways for Advancing Cervical Cancer Equity
- Decentralize Care: Task-shift outreach to community health volunteers (FCHVs) to overcome sociocultural stigma and increase screening uptake by nearly 50%.5
- Adopt Appropriate Tech: Utilize low-cost, portable isothermal POC assays (AmpFire) and self-sampling to reach remote populations at a fraction of traditional screening costs.15
- Support Vaccine Sovereignty: Facilitate the integration of affordable, indigenous vaccines like India’s Cervavac into national programs to ensure long-term primary prevention.27
Frequently Asked Questions About Cervical Cancer Prevention and Disparities
Why is the screening rate so low in India and Nepal?
Inaccessible primary care, geographic barriers, and sociocultural stigma regarding reproductive health are the primary drivers.7 Many women also lack the autonomy to seek care without familial consent.
What is the impact of the most recent, new HPV vaccination campaigns?
India's 2026 campaign targets 1.15 crore girls aged 14, providing free vaccines at government facilities to drastically reduce future incidence.4 Following the success of its 2025 multi-age campaign which reached 1.46 million girls (94% coverage), Nepal has officially integrated the HPV vaccine into its national routine immunization schedule as of February 2026, targeting approximately 350,000 girls in the 10-year-old cohort annually.
How do virtual tumor boards improve care in Nepal?
They allow local doctors in Janakpur to consult world-class experts from the US on complex cases, ensuring evidence-based treatment without requiring patients to travel internationally.
Can cervical cancer actually be eliminated?
While complete elimination (<4 cases per 100,000) is a long-term goal, meeting the WHO's 90-70-90 targets by 2030 will save millions of lives and put the world on the path to elimination by 2120.1
Author Bios
Suchit Shashikumar, MBBS

Dr. Suchit Shashikumar is an India-based physician and International Medical Graduate ranking in the top 10% of his cohort at BIMS, Karnataka. With over 20 weeks of U.S. clinical experience at premier institutions like UPMC, OSU & MSU, he brings a global perspective to patient care. Currently, he bridges clinical practice with systemic impact as a Research Assistant for Binaytara, where he spearheads oncology projects and grant development. A staunch advocate for health equity, Suchit also serves as a Fractional CMO focusing on rural healthcare access. When the stethoscope comes off, he’s either navigating rugged trails on his bike or masterminding elaborate photo-based scavenger hunts.
Jin Mou, MD, MSc, MPH, PhD

Dr. Jin Mou is the Implementation Science & Research Development Officer at Binaytara and is the first core member leading the development of the Binaytara Cancer Research Institute. Dr. Mou received her medical training from West China University of Medical Sciences, and Ph.D. in Public Health in Hong Kong. She has broad training in public health, evidence-based health care, clinical epidemiology, and health economics. Dr. Mou’s research spectrum extends from migrant heath, emerging infectious diseases to chronic diseases, patient-centered outcomes, and screening of complex chronic conditions including cancer. She has studied breast cancer screening at Cambridge University, UK, and served on the Hong Kong Breast Cancer Screening Review Board.
References
- World Health Organization. Cervical cancer fact sheet. Accessed March 16, 2026. https://www.who.int/news-room/fact-sheets/detail/cervical-cancer
- World Health Organization. Global Strategy to Accelerate the Elimination of Cervical Cancer. Geneva, Switzerland: World Health Organization; 2020.
- Duke Kunshan University. Global Action Plan for Cervical Cancer Elimination. Innovation Lab for Vaccine Delivery Research. Published 2026.
- Press Information Bureau. National rollout of HPV vaccination campaign. Government of India. Accessed March 16, 2026. https://www.pib.gov.in/PressReleseDetailm.aspx?PRID=2234009
- Shrestha AD, et al. Effect of a female community health volunteer-delivered intervention to increase cervical cancer screening uptake in Nepal: a cluster randomized controlled trial. Int J Environ Res Public Health. 2022;19(18):11636. doi:10.3390/ijerph191811636
- Centers for Disease Control and Prevention. Cervical cancer is preventable. Published 2020. Accessed March 16, 2026.
- Bray F, Laversanne M, Weiderpass E, Soerjomataram I. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2024;74(3):229-263. doi:10.3322/caac.21834
- OECD. Health at a Glance 2025: OECD Indicators. OECD Publishing; 2025. Accessed April 3, 2026. https://www.oecd.org/en/publications/health-at-a-glance-2025_15a55280-en.html
- International Institute for Population Sciences (IIPS) and ICF. National Family Health Survey (NFHS-5), 2019-21: India. IIPS; 2021.
- ResearchGate. Visual Inspection with Acetic Acid (VIA) Screening Program: 7 Years Experience in Rural South India. Published 2026.
- Shrestha S, et al. Cervical cancer screening seeking behavior among female community health volunteers of Surkhet District. medRxiv. Preprint published online July 25, 2024. doi:10.1101/2024.07.25.24311012
- ICO/IARC Information Centre on HPV and Cancer. Fact Sheet: HPV and Related Cancers in Nepal. Published 2023.
- United Nations Development Programme. Cold chains, code, and communities: how digital innovation is making India’s HPV vaccine work for every girl. Published March 16, 2026. Accessed April 6, 2026. UNDP India: https://www.undp.org/india/blog/cold-chains-code-communities-how-digital-innovation-making-indias-hpv-vaccine-work-every-girl
- World Health Organization Southeast Asia. Post introduction evaluation of HPV & TCV vaccination in Nepal December 2025. Published February 25, 2026.
- Karin, et al. "Point-of-Care Molecular Test for the Detection of 14 High-Risk Genotypes of Human Papillomavirus in a Single Tube." PMC. 2026; PMC12884375..
- Wei H, et al. Comparison of the ScreenFire and Xpert HPV assays for the detection of human papillomavirus and cervical precancer among women living with HIV in Malawi. J Med Virol. 2024;96(4). doi:10.1002/jmv.29545
- Paskett ED, et al. Impact of patient navigation interventions on timely follow-up for abnormal cervical screening. J Womens Health (Larchmt). 2016;25(1):15-21. doi:10.1089/jwh.2015.5323
- Nelson HD, Cantor A, Pappas M, Fu R. Patient Navigation Services for Breast and Cervical Cancer Screening and Follow-Up: A Meta-Analysis. JAMA Intern Med. 2025;185(15).
- Binaytara. Cancer care | global oncology programs. Published 2026. https://binaytara.org/global-oncology
- Cervical Cancer Action for Elimination. Cervical Cancer Elimination. CCAE; 2026.
- Prem K, et al. Evidence-based impact projections of single-dose human papillomavirus vaccination in India: a modelling study. Lancet Oncol. 2022;23(11):1419-1428. doi:10.1016/S1470-2045(22)00543-5
- Editorial. Cervical cancer: a disease of political will. Lancet Oncol. 2020;21(11):1391. doi:10.1016/S1470-2045(20)30594-0.
- Global determinants of cervical cancer outcomes: a health systems analysis. BMJ Oncology. 2026;5(1):e000975.
- Bridging the 'Know-Do' Gaps in Five Non-Communicable Diseases Using a Common Framework Driven by Implementation Science. PMC. 2023;10320809. [Updated for 2025/2026 context].
- New one-hour, low-cost HPV test could transform cervical cancer screening in Africa and beyond. ecancer. 2025;27093.
- Bringing Cervical Cancer Screening Closer to Women: Feasibility of Artificial Intelligence and Remote Assessment in Primary Health Care. Int J Public Health. 2026;1609094.
- Carpediem IAS. Indian HPV vaccine (Cervavac) and single-dose study: explained. Published 2026.
- UNICEF Nepal. Protecting girls against cancer. Published February 25, 2026. Accessed April 6, 2026.

Author
Jin Mou, MD, MSc, MPH, PhD
Binaytara
Implementation Science & Research Development Officer
View ProfileCategories
Share Article





