
AN AUTHORITATIVE RESOURCE FOR EVERYTHING ABOUT CANCER
Bridging the Access Divide in Relapsed DLBCL: Lessons from James’s Journey

Professor of Medicine
Improving Access to CAR-T and Bispecific Therapies in Relapsed DLBCL
Key Takeaways
- Timing is Critical: In relapsed DLBCL, the speed of treatment delivery is as important as the therapy itself.
- Bispecifics as a Bridge: "Off-the-shelf" bispecific antibodies may provide an immediate option for patients who cannot wait for CAR-T manufacturing.
- Coordination is Key: Success requires a shared-care model between community oncologists and specialty centers.
- Systemic Reengineering: Breakthroughs must focus on reducing geographical and socioeconomic barriers to ensure equitable utilization.
Introduction: Understanding Relapsed Diffuse Large B-Cell Lymphoma
Diffuse large B-cell lymphoma (DLBCL) is an aggressive but often curable disease. While frontline chemoimmunotherapy puts many patients into remission, the challenge intensifies for those who relapse. Recent advancements in chimeric antigen receptor T-cell (CAR-T) therapy and bispecific antibodies have fundamentally changed the prognosis for relapsed or refractory (R/R) large B-cell lymphomas. However, a significant gap remains between scientific innovation and patient access. This case highlights the clinical complexities of navigating advanced immunotherapies and the systemic barriers that determine who receives these life-saving treatments.
Case Presentation: Navigating Relapsed DLBCL in the Real World
Referral to a CAR-T Center
James, a 62-year-old accountant, was diagnosed with DLBCL and initially responded well to frontline treatment at his community cancer center. However, his lymphoma returned six months after completing therapy. His subsequent referral to an NCI cancer center introduced him to CAR-T therapy, now a standard of care for most patients with R/R large B-cell lymphomas (Westin & Sehn, 2022).
The complexity of CAR-T represents hope, but timing is critical (Figure 1). Disease progression during the manufacturing process can render patients ineligible. James was fortunate to receive CAR-T through a highly coordinated model:
Community Management: His local oncologist managed bridging therapy and labs.
Specialty Center: The CAR-T center managed lymphodepleting chemotherapy and administration.
Navigation: An insurance case manager and center navigator facilitated scheduling, approvals, and caregiver education.
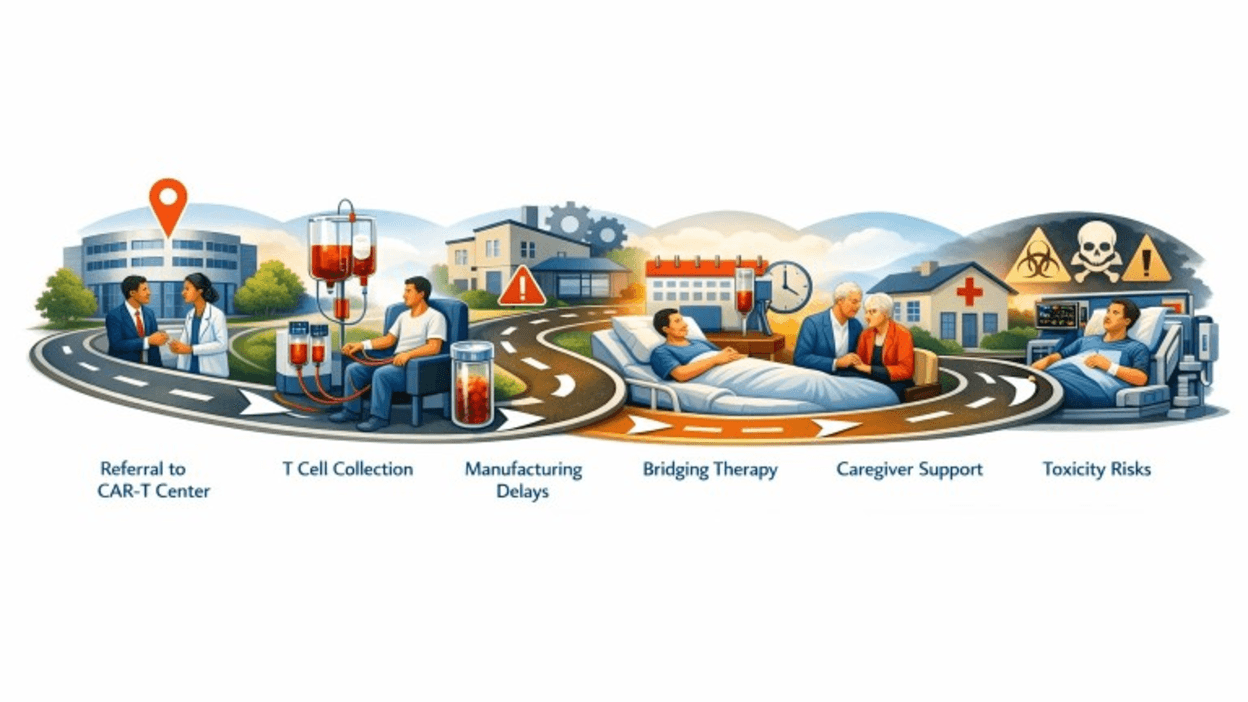
Figure 1: Complex CAR-T journey
When CAR-T Response Is Incomplete: The Role of Bispecific Antibodies
Transition to Bispecific Antibodies when James’s response to CAR-T was incomplete, his oncologist transitioned him to CD20/CD3 bispecific antibodies, a class of therapies reshaping lymphoma care (Bock & Epperla, 2025; Shumilov et al., 2025). Unlike CAR-T, bispecifics are "off-the-shelf," requiring no cell collection or manufacturing delay, an essential factor for patients with fast-moving disease.
Despite the clinical efficacy, James faced logistical "hidden costs." Initial doses required extended monitoring two hours from home, necessitating time off work, transportation planning, and significant caregiver reliance. While James achieved complete remission, his success was supported by good insurance, a motivated referring oncologist, strong caregiver support, and adequate financial reserves—factors not available to all patients.
Key Clinical Lessons from Relapsed DLBCL Care
James’s experience highlights that in R/R lymphoma, speed matters. Biology can accelerate quickly, and delays translate directly into worse outcomes despite the availability of novel therapies (Battiwalla et al., 2025).
The Lymphoma Access Divide
Lymphoma care exposes access inequities with unusual clarity (Kansagra et al., 2020). Currently, only about one-third of patients who would benefit from these treatments are able to receive them (Figure 2). Five systemic barriers repeatedly surface:
- Geography: CAR-T and early bispecific treatments are concentrated in tertiary centers, often far from a patient's home.
- Time: Referral and insurance delays collide with aggressive disease kinetics.
- Fitness and Eligibility: Delays worsen performance status, rendering patients ineligible for intensive therapies.
- Socioeconomic Strain: Caregiver requirements and travel disproportionately exclude vulnerable patients.
- Fragmented Care: A lack of awareness among community oncologists and poor coordination between community practices and specialty centers leads to lost opportunities.

Figure 2: Access funnel with barriers for relapsed DLBCL
Redesigning Care Delivery to Expand Access to Advanced Lymphoma Therapies
Bispecific antibodies and CAR-T have fundamentally changed outcomes in relapsed lymphoma. For the first time, patients failing chemotherapy still have meaningful, potentially durable options. Scalable solutions have been proposed by or are in the process of being implemented by different stakeholder groups to ensure that these advances are able to change the landscape for relapsed lymphoma:
- Early referral at first relapse: Community outreach and education by advanced therapy centers to increase awareness about ‘not waiting for multiple recurrences before referring the patient’ and encouraging bidirectional communication, along with the use of standardized referral templates and automatic intake triage, can help increase timely access. Pharmaceutical companies can enhance outreach efforts, though it might be for specific products.
- Hub-and-spoke lymphoma networks: Many advanced therapy centers are beginning to establish a network of community practices to expand access to these therapies through a shared care model with shared order sets and toxicity escalation protocols for routine patients who can be treated at the local center, while steering the more complex patients to the academic center.
- Standardization of financial, operational, and clinical workflows: American Society for Transplantation and Cellular Therapy (ASTCT) 80/20 Taskforce has proposed standardization and streamlining of onboarding, educational requirements to reduce variability and expedite access (Nikiforow et al., 2023). Another multi-disciplinary panel of physician leaders and payers proposed streamlining of prior authorization processes and payer pathways for approval and reimbursement to help improve equitable access to these lifesaving therapies (Uttam Chandani & Khera, 2026).
- Virtual consultation and remote monitoring: This can help improve access to pre and post-cell therapy care, especially to patients in remote areas, while minimizing burden on the patient as well as reducing inpatient bottlenecks. While programs have reported on the feasibility and early success of the remote patient monitoring programs for patients undergoing CAR-T and Bispecifics, their impact on expanding access and reducing non-medical costs for patients needs to be systematically assessed.
- Resources for social support: Lodging and caregiver support are essential treatment-enabling infrastructure, and most academic centers have variable levels of support in terms of social worker availability and resources to help with these challenges, which disproportionately affect the underrepresented groups in the community. There is a need to grow these resources and spread awareness about them. This may require partnerships with pharma and patient advocacy organizations.
Expert Commentary and Insights: Innovation in Access Must Match Innovation in Treatment
The real breakthrough in lymphoma care will not come solely from a new molecular target or construct; it will come from reengineering care delivery processes. Innovation in treatment must be matched by innovation in access. It is important to measure the improvement in equity across the entire care continuum from diagnosis to referral/access to evaluation, treatment, and outcomes across population groups based on race/ethnicity, economic status, and geography. This requires societal will and multi-stakeholder efforts to ensure that innovation in treatment matches innovation in access.
The ACCESS Initiative, a collaborative effort of ASTCT and the National Marrow Donor Program (NMDP), serves as a model for multi-stakeholder efforts to alleviate outcome disparities in the hematopoietic cell transplantation (HCT) and cell therapy (CT) ecosystem (Team et al., 2022). It is a call to action for every stakeholder (Figure 3) to measure and improve equity across the entire care continuum—from diagnosis to treatment and long-term outcomes.
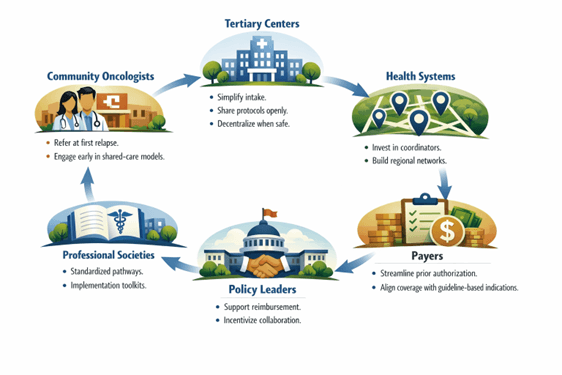
Figure 3: Role of individual stakeholders in improving access to advanced therapies
Patient Perspective: The Hidden Costs of Accessing Advanced Therapies
For patients like James, advanced therapies are a lifeline, but the burden of accessing them is high. The "hidden costs" of travel, meals, and lost income can accumulate quickly, making a "curable" disease feel out of reach for those without a robust support system. Ensuring that innovation does not lead to further exclusion requires a focus on quality-of-life and equity-driven care models.
Frequently Asked Questions About CAR-T and Bispecific Antibodies
Why is CAR-T therapy considered "complex" compared to other treatments?
CAR-T requires a personalized manufacturing process where a patient’s T-cells are collected, re-engineered, and expanded. This can take weeks, during which the lymphoma may progress.
What makes bispecific antibodies different from CAR-T?
Bispecific antibodies are "off-the-shelf," meaning they are ready for immediate use. They do not require cell collection or manufacturing time, making them ideal for rapidly progressing diseases.
How can community oncologists improve access for their patients?
Early referral at the first sign of relapse and establishing "hub-and-spoke" relationships with specialty centers can ensure patients are evaluated while they are still healthy enough for treatment.
About Author
Nandita Khera is a Professor of Medicine at the Mayo Clinic College of Medicine and a Consultant in the Division of Hematology/ Oncology at Mayo Clinic Arizona. She treats patients with hematological malignancies and some solid tumors, especially those needing blood and marrow transplants (BMT) and/or cell therapy (CT). Her research interests include health economics, patient-reported outcomes, disparities in care, and dissemination/ implementation of research into practice in cancer. She has published several papers on outcomes and quality of care in cancer patients. She holds leadership positions on the various committees in organizations/ societies in hematology and BMT. She helps lead the patient-targeted efforts towards eliminating barriers to transplant and cell therapy as part of the ASTCT-National Marrow Donor Program's ACCESS Initiative. She is on the board of directors of ENGRAFT: a learning health network that strives to improve the lives of patients undergoing BMT/CT by bringing together all relevant stakeholders. She is the chair of the Therapeutic Strategy subcommittee at Mayo Clinic, where she works with a diverse team to bring transformative therapies to practice while ensuring financial stability for the institution. She teaches classes as part of the high value cost conscious care curriculum at Mayo Medical School and is a faculty member at large in the Science of Health Care Delivery (SHCD) Curriculum Subcommittee.
Works Discussed
- Battiwalla, M., Tees, M., Flinn, I., Pantin, J., Berdeja, J., Gregory, T., Maris, M., Bhushan, V., Vance, E., Mathews, J., Bachier, C., Shaughnessy, P., Ramakrishnan, A., Malik, S., Mori, S., Martin, C., Billups, R., Blunk, B., LeMaistre, C. F., & Majhail, N. S. (2025). Access barriers to anti-CD19+ CART therapy for NHL across a community transplant and cellular therapy network. Blood Advances, 9(2), 429-435. https://doi.org/10.1182/bloodadvances.2024014171
- Bock, A. M., & Epperla, N. (2025). Therapeutic landscape of primary refractory and relapsed diffuse large B-cell lymphoma: Recent advances and emerging therapies. Journal of Hematology & Oncology, 18(1), 68. https://doi.org/10.1186/s13045-025-01702-5
- Kansagra, A., Farnia, S., & Majhail, N. (2020). Expanding Access to Chimeric Antigen Receptor T-Cell Therapies: Challenges and Opportunities. American Society of Clinical Oncology Educational Book(40), e27-e34. https://doi.org/10.1200/EDBK_279151
- Nikiforow, S., Frigault, M. J., Frey, N. V., Gardner, R. A., Komanduri, K. V., Perales, M. A., Kebriaei, P., Warkentin, P. I., Pasquini, M., Aho, J. L., Levine, B. L., Heslop, H. E., Hlucky, T. L., Habucky, K., Gharibo, M., Jagasia, M., & Locke, F. L. (2023). Paving the Road for Chimeric Antigen Receptor T Cells: American Society for Transplantation and Cellular Therapy 80/20 Task Force Consensus on Challenges and Solutions to Improving Efficiency of Clinical Center Certification and Maintenance of Operations for Commercially Approved Immune Effector Cell Therapies. Transplant Cell Ther, 29(4), 228-239. https://doi.org/10.1016/j.jtct.2023.01.021
- Shumilov, E., Scholz, J. K., Seib, M., Mazzeo, P., Wurm-Kuczera, R., Vucinic, V., Holtick, U., Boyadzhiev, H., Melchardt, T., Hölscher, A., Schultze-Florey, C., Abdelhafez, A., Velazquez, G. F., Ossami Saidy, A., Lesan, V., Schnetzke, U., Kerkhoff, A., Bacher, U., Ghandili, S., . . . Lenz, G. (2025). Outcomes of bispecific antibody therapy after CAR T-cell failure in relapsed/refractory large B-cell lymphoma. Blood Advances, 9(15), 3955-3966. https://doi.org/10.1182/bloodadvances.2024015719
- Team, A. W., Auletta, J. J., Sandmaier, B. M., Jensen, E., Majhail, N. S., Knutson, J., Nemecek, E., Ajayi-Hackworth, F., & Davies, S. M. (2022). The ASTCT-NMDP ACCESS Initiative: A Collaboration to Address and Sustain Equal Outcomes for All across the Hematopoietic Cell Transplantation and Cellular Therapy Ecosystem. Transplantation and Cellular Therapy, Official Publication of the American Society for Transplantation and Cellular Therapy, 28(12), 802-809. https://doi.org/10.1016/j.jtct.2022.09.020
- Uttam Chandani, K., & Khera, N. (2026). Bridging the Gap: Identifying and Overcoming Barriers to CAR-T Access Through Streamlined and Standardized Pathways. Mayo Clin Proc Innov Qual Outcomes, 10(1), 100693. https://doi.org/10.1016/j.mayocpiqo.2025.100693
- Westin, J., & Sehn, L. H. (2022). CAR T cells as a second-line therapy for large B-cell lymphoma: a paradigm shift? Blood, 139(18), 2737-2746. https://doi.org/10.1182/blood.2022015789





